|
The algorithms that calculate the isoelectric point of proteins and peptides use the [enderson Hasselbalch equation, and believe it or not, we have a henderson hasselbalch calculator that will tell you all about it. Protein electrophoresis – A test that measures the amount of a specific protein, in the blood, based on its net electrical charge.Ion-exchange chromatography – Chromatographic separation method that separates proteins in a solution/environment based on their net charge and.It helps in methods used for protein separation like: The knowledge of isoelectric points is significant in protein studies. The acidic amino acids have pI values between a pH of 5.0 to 7.0, while the basic amino acids have a higher pH to attain their isoelectric point. Let's look at the isoelectric points of specific biomolecules.Īmino acids are zwitterions when they attain their isoelectric point. It tells us when a particular molecule has attained the electrically neutral state, and the pI can affect the solubility of said molecule. Just follow the titration scheme of the amino acid whose pI u want to calculate and form the scheme identify the zwitterionic form of amino acid and then. The isoelectric point is an essential topic in chemistry and biochemistry. At pH values above or below the isoelectric point, the molecule will have a net charge which depends on its pI value as well as the pH of the solution in which the amino acid is found. Each amino acid has its own pI value based on the properties of the amino acid. As long as you have any two of the three values to input, the calculator will work fine for you. The zwitterion of an amino acid exists at a pH equal to the isoelectric point. Approach 1 deprotonate e 3 (i.e. Let's call the ends e 1, e 2 and e 3 (from left to right). 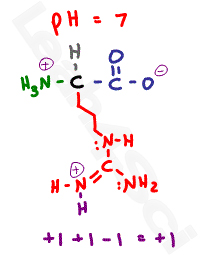
Remember, our tool works in reverse and any other order you want to. Since the p I is the p H at which the amino acid has no overall net charge, you need to average the p K a values relevant to the protonation/deprotonation of the form with no net charge. Input these values in the calculator and the result is 6.55 6.55 6.55 Say you have molecule X, its p K a = 3.7 pKa = 3.7 p K a = 3.7 and p K b = 9.4 pKb = 9.4 p K b = 9.4. 
This value indicates the pH at which your molecule carries no net electric charge. The result is that molecule's isoelectric point (pI value).Now, coming back to the point at hand, how to use the isoelectric point calculator? Follow the given steps, and that's it: For an amino acid with a neutral side chain, only the amine. The dissociation constant represents the capability of a substance to dissociate into ions in a solution. The isoelectric point is the pH where the amino acid is a zwitterion, or carries no overall charge. 
What are pKa and pKb values, you might wonder? These are the dissociation constants of acids and bases. It determines the isoelectric point of molecules based on their pKa and pKb values. This is called the isoelectric point of the amino acids, and is designated. Our isoelectric point calculator is a tool you would love to use on the go. At some intermediate pH, the amino acid is a zwitterions, and carries no net charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
